ISO 9001 holders looking at Article 17 of the EU AI Act (Regulation (EU) 2024/1689) are not starting from zero. A well-implemented ISO 9001 system already covers the structural foundations Article 17 demands: documented procedures, change control, internal audit, corrective action, and resource management. Most product manufacturers with a mature QMS satisfy 60–70% of what the AI Act quality management system requires. The gap is about extending into territory ISO 9001 was never designed to reach. Three areas require genuinely new procedures: AI-specific risk management, post-market monitoring with structured feedback loops under Article 72, and the AI technical documentation under Article 11. High-risk AI obligations currently apply from 2 August 2026 for Annex III systems. A Digital Omnibus proposal could push this to 2 December 2027 if adopted. Either way, conformity assessment preparation takes months, the window to act is now.
What Does Article 17 Actually Require?
Article 17 of Regulation (EU) 2024/1689 requires providers of high-risk AI systems to establish, document, implement, and maintain a quality management system. The system must be proportionate to organisational size, a concession that gives SMEs flexibility without reducing substantive requirements. Coverage must span the full AI lifecycle, from initial design through to post-market monitoring.
Article 17 lists thirteen specific areas the QMS must address. These cover the regulatory compliance strategy; design and development procedures; testing, verification, and validation; technical specifications; data management; and the risk management system under Article 9. Post-market monitoring under Article 72, incident reporting under Article 73, communication procedures, record-keeping under Article 18, resource management, and an accountability framework complete the list. Article 17(3) and (4) confirm that providers already subject to QMS obligations under other EU legislation may integrate Article 17 into their existing system. Medical device manufacturers, for instance, do not need a parallel QMS, they extend the one they already operate.

Note: The draft harmonised standard for Article 17 compliance is prEN 18286, under development within CEN/CENELEC. A detailed analysis of its clause structure and mapping to Article 17 by CMS Law-Now covers the October 2025 public enquiry version. Until publication, ISO/IEC 42001 remains the closest available reference, but neither standard removes the obligation to address the Article 17 requirements directly.
How Article 17 Maps to ISO 9001: Where the Overlap Holds
Understanding which parts of your existing system translate cleanly saves time and avoids unnecessary rework. The mapping below focuses on the clauses that matter most.
ISO 9001 provides solid coverage for several Article 17 requirements. Clause 4 (Context of the organisation) maps to the regulatory compliance strategy, scoping the QMS around internal and external issues already forms this foundation. Design, development, testing, and validation under Clause 8 (Operation) extend reasonably to AI system work, though AI Act traceability demands are more granular. Nonconformity and corrective action under Clause 10.2 maps directly to incident reporting and corrective actions under Articles 20 and 73.
Two clauses only partially cover their Article 17 counterparts. Clause 6.1 (Actions to address risks and opportunities) touches the risk management requirement. It was not written for AI hazards such as training data bias, distributional shift, or algorithmic drift.
Clause 9.1 (Monitoring, measurement, analysis, and evaluation) provides the process foundation for post-market monitoring, but Article 72’s data collection and feedback obligations go much further.
Three areas have no ISO 9001 equivalent at all. First is the AI-specific risk management procedure under Article 9. Second is the structured feedback mechanism under Article 72, linking field data back into the technical documentation. Third is EU database registration under Articles 49 and 71. These three gaps define the three workstreams of your extension project.
Building the AI-Specific Risk Management Procedure
Article 9 demands a continuous, iterative risk management process spanning the entire AI system lifecycle. A one-time pre-market assessment does not satisfy it.
As the EU AI Act risk pyramid explains, high-risk classification triggers the full Chapter III obligation set, including this integrated risk management system. Your Article 9 procedure must cover four areas. First, identifying risks to health, safety, and fundamental rights from the AI system’s intended use. Second, evaluating severity and likelihood of those risks. Third, implementing controls such as algorithmic safeguards, human oversight requirements, and performance thresholds. Fourth, verifying those controls remain effective over time.
The procedure must address AI hazard categories absent from conventional product risk work. Training data risks cover representativeness, bias, and labelling accuracy. Performance risks cover accuracy degradation and distributional shift, cases where the system encounters inputs that differ statistically from its training data. Human oversight risks cover decision scenarios where no qualified person can intervene adequately. Each category must link to the technical documentation under Article 11, keeping risk assessment and technical file consistent.
Tip: EN ISO/IEC 23894 (Guidance on risk management for AI systems) maps reasonably well to Article 9 and can serve as the procedural basis. CEN/CENELEC lists it as a non-harmonised standard relevant to AI Act compliance, alongside EN ISO/IEC 23053 and EN ISO/IEC 8183.
Two downstream documents draw from this procedure. The technical file under Article 11 records controls and residual risks. Your post-market monitoring plan under Article 72 then defines the field indicators that signal emerging risk.
Post-Market Monitoring Under Article 72: What Your QMS Must Provide
Article 72 sets post-market monitoring obligations more specific than anything in ISO 9001. Providers must establish a system that actively and systematically collects, documents, and analyses performance data throughout the AI system’s lifetime. The monitoring plan must form part of the technical documentation, making it a living document, not a post-launch addition.
This requirement creates four process gaps most existing QMS do not address. First, a data collection mechanism. The procedure must specify what data to collect, from whom, deployers under Article 26(5), automatic logs under Article 12, or other sources, and at what frequency. Second, an analysis procedure: collected data gets evaluated against the performance thresholds and risk indicators from the Article 9 assessment. Third, a feedback loop: when monitoring signals a performance change or emerging risk, the technical file must be updated and the risk assessment reviewed. Fourth, an escalation path. Serious incidents under Article 3(49) — involving death, serious harm, or critical infrastructure disruption, require reporting to the relevant market surveillance authority under Article 73.
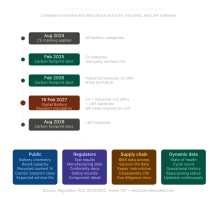
Article 73 sets reporting timelines at 15 days for serious incidents, 10 days where death is involved, and 2 days for widespread infringements. Your incident procedure must reflect these timelines and name the competent authority for each deployment Member State.
Note: The Commission must publish an implementing act specifying a post-market monitoring plan template. At the time of publication, it has not appeared in the Official Journal. Monitor the Commission’s AI Act standardisation page for updates, as the template may impose format requirements once adopted.
Products already regulated under a sectoral framework may benefit from integration. Medical devices under Regulation (EU) 2017/745, or machinery under Regulation (EU) 2023/1230, can merge their AI post-market monitoring obligations into the existing post-market surveillance system, provided equivalent safeguards apply. For a broader view of how the AI Act interacts with other concurrent obligations, see the AI Act vs Cyber Resilience Act comparison. It illustrates how overlapping frameworks can share infrastructure rather than duplicate it.
EU Database Registration Under Articles 49 and 71
Registration in the EU database under Article 71 is a pre-market obligation, not an ongoing administrative task. Article 49(1) requires providers of Annex III high-risk systems to register themselves and their system before market placement or service deployment.
Annex VIII, Sections A and B, specifies what each entry must contain. Required fields include provider identity and contact details, intended purpose, the Annex III classification category, deployment Member States, conformity assessment route, CE marking details, and post-market monitoring contact. Every entry must stay current. A new deployment territory, a change in conformity assessment route, or a substantial modification each require an update to the record.
The QMS must assign clear ownership of the registration entry. Compliance or legal typically holds this responsibility, but the underlying data, system description, intended purpose, conformity assessment status, comes from R&D and quality functions. Your procedure should define who registers, who can update, and how relevant changes from development or monitoring reach the person holding the database entry.
Tip: Most Annex III registration entries are publicly accessible. Competitors, customers, and market surveillance authorities can all view your system’s registration status. Treat the entry as a CE marking declaration, a public statement of conformity, not an internal record.
The Technical Documentation Gap: Beyond the Standard Product Technical File
The AI technical documentation under Article 11 and Annex IV will feel structurally familiar to anyone who builds CE product technical files. The content, however, goes significantly further.
A conventional technical file contains a product description, applied standards, test reports, a risk assessment, and the Declaration of Conformity. Article 11 requires all of that, plus additional AI-specific content. This includes a description of the AI system’s components and development process. It also covers training methodology and datasets with the criteria for their selection, intended performance metrics and achieved test results, the post-market monitoring plan, and a description of any predetermined changes that may affect conformity post-deployment.
That last requirement is critical for machine learning systems that update in the field. The technical documentation must define the boundaries within which post-deployment updates remain permissible without triggering a new conformity assessment. Updates outside those boundaries may constitute a substantial modification under Article 83, requiring re-assessment before the modified version ships.
This point is one of the sharpest differences between AI compliance and the electrical safety and EMC frameworks most product engineers know. As the engineer’s guide to AI certification on this site explains, the regulation must account for systems that change after they leave the factory — something no electrical safety standard has ever needed to do.
QMS Readiness: Ten Questions Before Conformity Assessment
Before submitting a high-risk AI system for conformity assessment, a compliance manager must be able to answer these ten questions affirmatively. Together, they cover the minimum substantive requirements of Articles 9, 11, 17, 72, 49, and 71.
Has the QMS been formally scoped to include the AI system? Does an Article 9 risk management procedure exist with a current risk assessment for the system? Is the Annex IV technical documentation complete, training data description, performance metrics, and monitoring plan included? Are predetermined change boundaries defined for any system that learns or updates post-deployment? Does the post-market monitoring system operate with defined data collection mechanisms and analysis triggers? Do incident reporting thresholds and timelines align with Article 73? Has EU database registration under Article 49 been completed? Does the registration record have an assigned owner with update authority? Has the QMS been audited specifically against Article 17 — not only against ISO 9001? Are the Declaration of Conformity under Article 47 and CE marking procedure under Article 48 in place and linked to the QMS documentation?
Every negative answer is a gap. Any gap is a delay to market placement. Where the system is already in service past the applicable deadline, it becomes a non-compliance. Fines reach up to €15 million or 3% of global annual turnover under Article 99(3). For the latest on applicable deadlines and any Digital Omnibus changes, the Commission’s AI Act standardisation page is the most current reference.
Frequently Asked Questions
Does ISO 9001 certification satisfy Article 17 of the EU AI Act? No, but it provides a strong foundation. ISO 9001 covers structural and procedural elements that Article 17 requires. Training data governance, algorithmic risk management, and post-market monitoring of model performance all fall outside its scope. Those gaps require explicit QMS extensions.
When does the Article 17 QMS obligation apply to high-risk AI systems? For Annex III systems, the current obligation date is 2 August 2026. For safety components under Annex I legislation, the deadline is 2 August 2027. A Digital Omnibus proposal may extend the Annex III date to 2 December 2027 if adopted — monitor official sources before finalising your compliance timeline.
What is the difference between the Article 9 risk management system and a standard product risk assessment? A standard product risk assessment runs once before market placement. Article 9 demands a continuous, iterative process covering the full system lifecycle. It must address AI-specific hazard categories — training data bias, distributional shift, and human oversight failures. It must also link directly to the post-market monitoring plan and technical documentation.
Does the post-market monitoring plan need to be submitted before market placement? No pre-submission is required. The plan must form part of the Annex IV technical documentation and be available to market surveillance authorities on request. Once the Commission publishes a mandatory template, compliance with that format will also be required.
Can a company integrate the AI Act QMS into an existing ISO 13485 or MDR quality system? Yes. Article 17(3) and (4) explicitly permit integration with QMS obligations under other EU legislation. The integrated system must demonstrably cover all Article 17 requirements — integration reduces administrative overhead, not substantive obligations.
Conclusion
For organisations with a mature ISO 9001 system, adapting to Article 17 is a focused extension project, not a rebuild. Structural foundations carry across. The genuine work lies in three areas. First, an Article 9 risk management procedure addressing AI-specific hazards. Second, a post-market monitoring system under Article 72 linking operational data back into the technical documentation. Third, the EU database registration workflow under Articles 49 and 71.
Deadlines are close. Conformity assessment takes time, and assessors cannot certify against a QMS that does not yet cover the AI-specific requirements. The compliance journey for high-risk AI follows the same logic as CE marking did when it became mandatory. It requires methodical documentation, safety as a design input, and no shortcuts after the fact.
Start with the classification question. For the full picture of what high-risk designation means and how it is determined, the EU AI Act risk pyramid is the right first step before any QMS extension work begins.